Independent Provider of Microscopy and Imaging Solutions in the Nordics
Visi4
CONTACT US
Info@Visi4.com
Tel: +45 61666619
Peter Thomsen
Innovation &
Microscopy expert.
Stengårds Alle 31A
2800 Lyngby
Denmark
PROFILE
Visi4 commercialize advanced imaging solutions.
These solutions are proven in Visi4's development of vascular implants.
Overview
Cardiovascular disease is the leading cause of death and disease globally causing over 17 million people dying each year. 1–6% of vascular graft procedures are infected. The costs associated with one infection are estimated at US$40,000. Mortality ranges between 13% and 58%, and the amputation rate in survivors varies between 8% and 52%. Immediate occlusive complications are seen in up to 18% of patients. Restenosis occurs around 10 years after the procedure.
Medical need
Most blood or tissue-contacting medical devices, including vascular grafts, are comprised of standard materials originally not designed for surgery. Therefore vascular access devices are flawed by a number of problems, resulting in frequent device thrombosis and infection. Blood proteins adsorb strongly to the surfaces of existing grafts which may promote coagulation. These grafts furthermore activate the complement system which intensifies coagulation through cross-talk reactions. Overall, the resulting graft thrombosis and fibrin sheath formation leads to flow problems and device failure. The fibrin sheath furthermore provides a habitable environment for bacteria.
Attempts to add a coating to the incompatible materials have never truly solved the problem. This recently made a key opinion leader, B.D. Ratner, ask the question: “why after 60 years of serious study of blood compatibility and development of blood compatible surfaces do we not have truly blood compatible surfaces?”
As a result, a substantial medical need exists for an approach that combines the benefits of high material biostability, high material hemocompatibility and a material capable of expanding in response to pulsating blood flow.
Innovation & Approach
An ideal vascular graft has the appropriate size to match the host blood vessels, allows for rapid healing, ease of handling, exhibits increased porosity, resistance to infection, low thrombogenicity and is low cost.
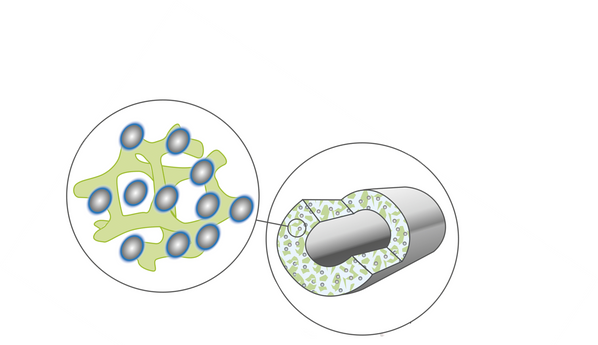
The Visi4 approach is unique. Thrombosis-resistant hydrogels are impregnated into a bulk silicone vascular graft material. The resulting interpenetrating polymer network (IPN) integrates hemocompatible properties into the silicone bulk material. The result is highly beneficial mechanical and hemocompatible properties in one flexible material which does not decompose in vivo.
In this way we leverage the blood vessel's natural design and function to redefine how cardiovascular diseases should be treated with vascular grafts.
FOLLOW Visi4